A Water Molecule Is Best Described as
Carbon dioxide C O 2 has D h symmetry a perfectly-symmetrical arrangement of C and O. A model of a water.
Water Structure Properties Molecule Physical Properties A Level
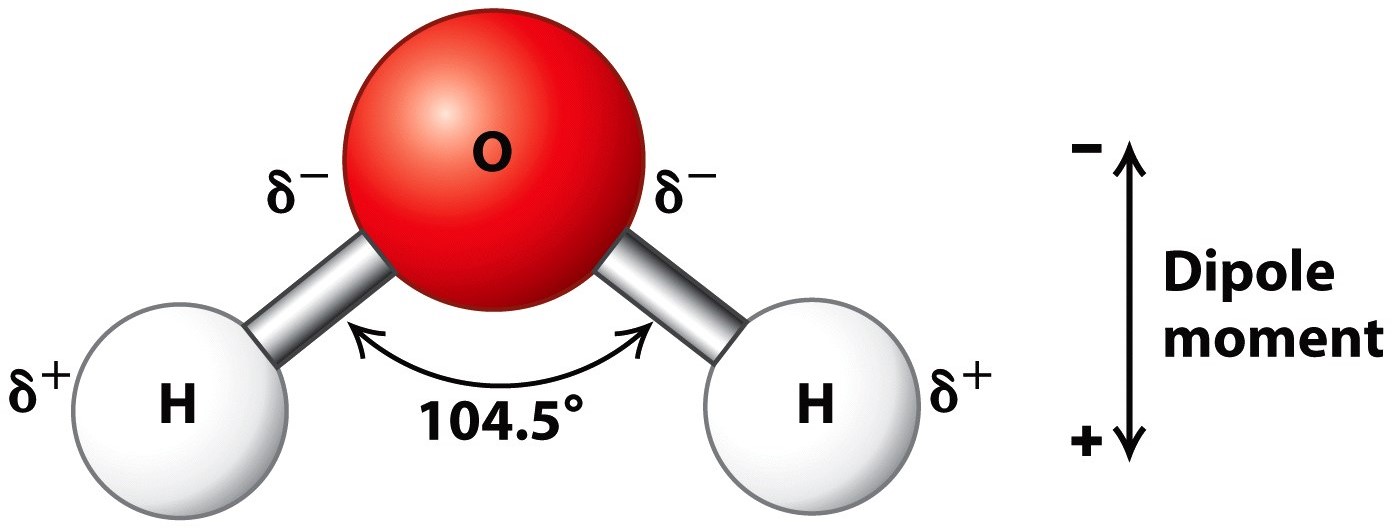
It is a polar molecule because its polar bonds are arranged asymmetrically.
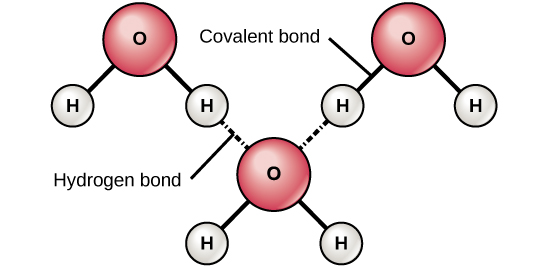
. A universal solvent capable of dissolving polar and non-polar moleculos. The following molecule is best described as a. A water molecule is made of two atoms of hydrogen and one atom of oxygen as.
Jfor10 jfor10 14052018 Biology Secondary School answered In a water molecule each bond between the oxygen atom and the two hydrogen atoms are best described as. A molecule of water B. Human history is determined by a series of supernatural events.
The Water Molecule Is Best Described as Ne_Weston166 April 13 2022 We dont longer stand behind the arguments and the validity of these claims Through the 1990s Dr. 2 The oxygen end is negative relative to the end with the two hydrogen atoms. Water is a source of _____ for chemical reactions in cells.
The Water Molecule Is Best Described as By Da_Meadow91 09 Apr 2022 Post a Comment Water Facts Properties Structure Compounds Summary Lesson Summary Water And Life Article Khan Academy Water Molecules And Their Interaction With Salt U S Geological Survey. If you need a better explanation just as in the comments. Advertisement Advertisement Brainly User Brainly User.
The molecule can have the same or different types of atoms. In a beaker of water the bonds within a water molecule can best be described as. The most common example of this is water.
Water is best described as A good solvent capable of dissolving non-polar molecules. Humans should help in the conservation of other animal species. In a water molecule each bond between the oxygen atom and the two hydrogen atoms are best described as.
Select the correct answer. A polar molecule where the hydrogen is more electronegative than oxygen A good solvent capable of dissolving polar molecules and ions. One red sphere attached to two white spheres Which phrase best describes the image.
Glucose is a source of energy which is produced by green plants with the help of carbon dioxide and water in the presence of sunlight. Masaru Emoto performed a series of experiments observing the physical effect of words prayers music and environment on the crystalline structure of water. Since oxygen is more electronegative as compared to hydrogen atoms the shared electrons are attracted towards the oxygen atom.
O single covalent bond double covalent bond triple covalent bond ionic bond. Dissolving is best described as. What is the diatomic nitrogen molecule best described as.
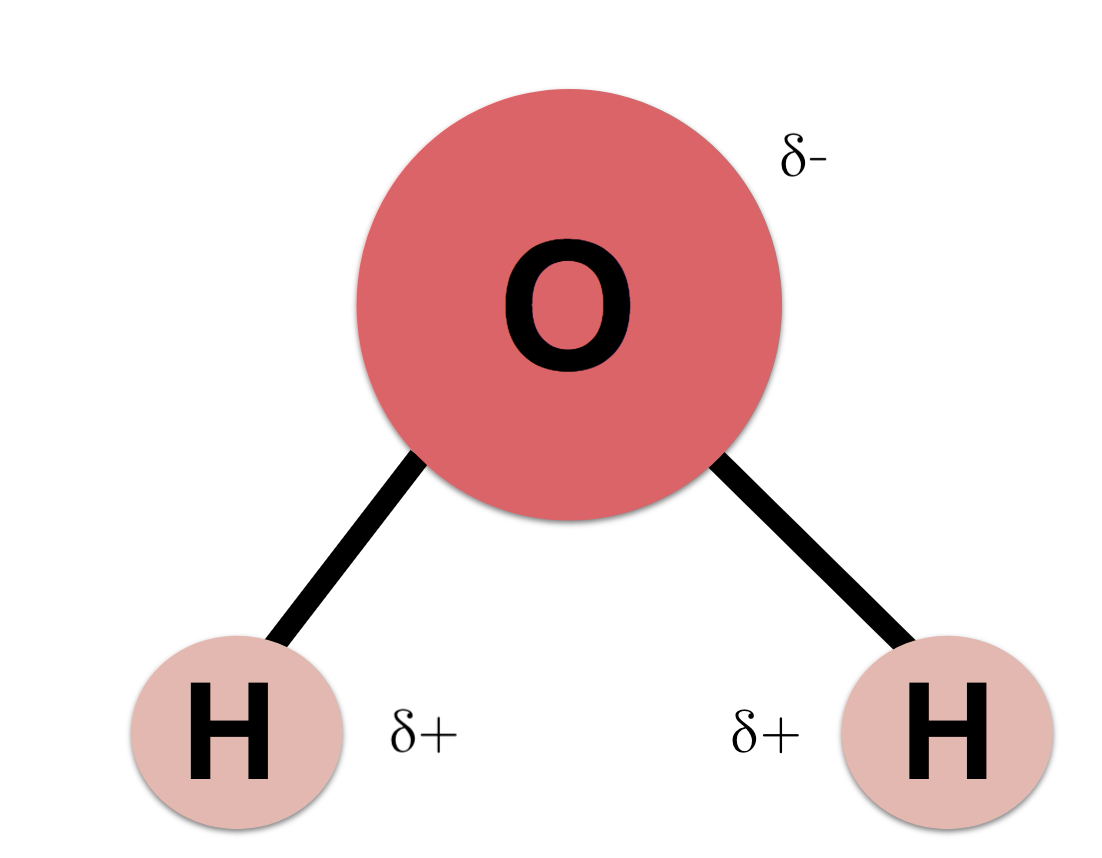
A water molecule consists of one oxygen atom bonded to two hydrogen atoms by covalent bonds. Water Is Polar There is no overall charge to a water molecule but there is a slight positive charge on each hydrogen atom and a slight negative charge on the oxygen atom. 1 The oxygen end is positive relative to the end with the two hydrogen atoms.
Is the molecule best described as an energy-rich molecule. Water is sometimes called the universal solvent. This is a form of stored energy which is then released when oxidation of glucose takes place.
A molecule is 2 or more atoms bonded together. View the full answer. Get the answers you need now.
A mingling of molecules andor ions. The unequal sharing of electrons within a water molecule makes the water molecule _____. Humans and bacteria share a common genetic code.
Which best describes how charges are distributed on a water molecule. Because of these charges. A water molecule is made of two atoms of hydrogen and one atom of oxygen as shown in the image.
The molecule that is best described as glucose has the chemical formula C6H12O6. A single non-polar covalent bond. 3 The charge is neutral and equal throughout the molecule.
Humans are controlled by forces beyond our understanding. A base unit of water C. This imparts partial negative charge to the oxygen atom and partial positive charge to hydrogen atoms.
Water H 2 O has C 2 v symmetry an asymmetric arrangement of O and H. The oxygen end is positive relative to the end with the two hydrogen atoms. Ionic bonds polar covalent bonds non-polar covalent bonds hydrogen bonds When a pair of electrons is shared between two atoms the bond is called.
The tendency of an atom to pull electrons toward itself is referred to as its _____.

Why Life Depends On Water Biology For Non Majors I
Lesson Summary Water And Life Article Khan Academy
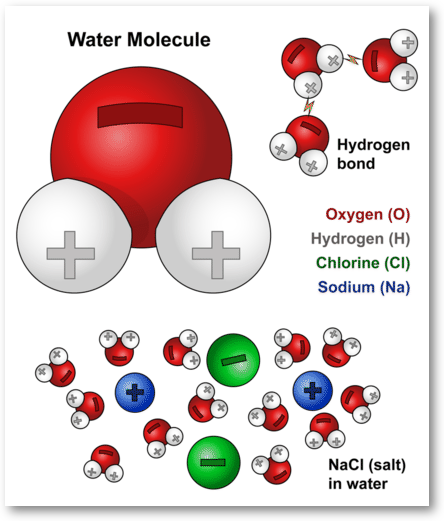
Water Molecules And Their Interaction With Salt U S Geological Survey
No comments for "A Water Molecule Is Best Described as"
Post a Comment